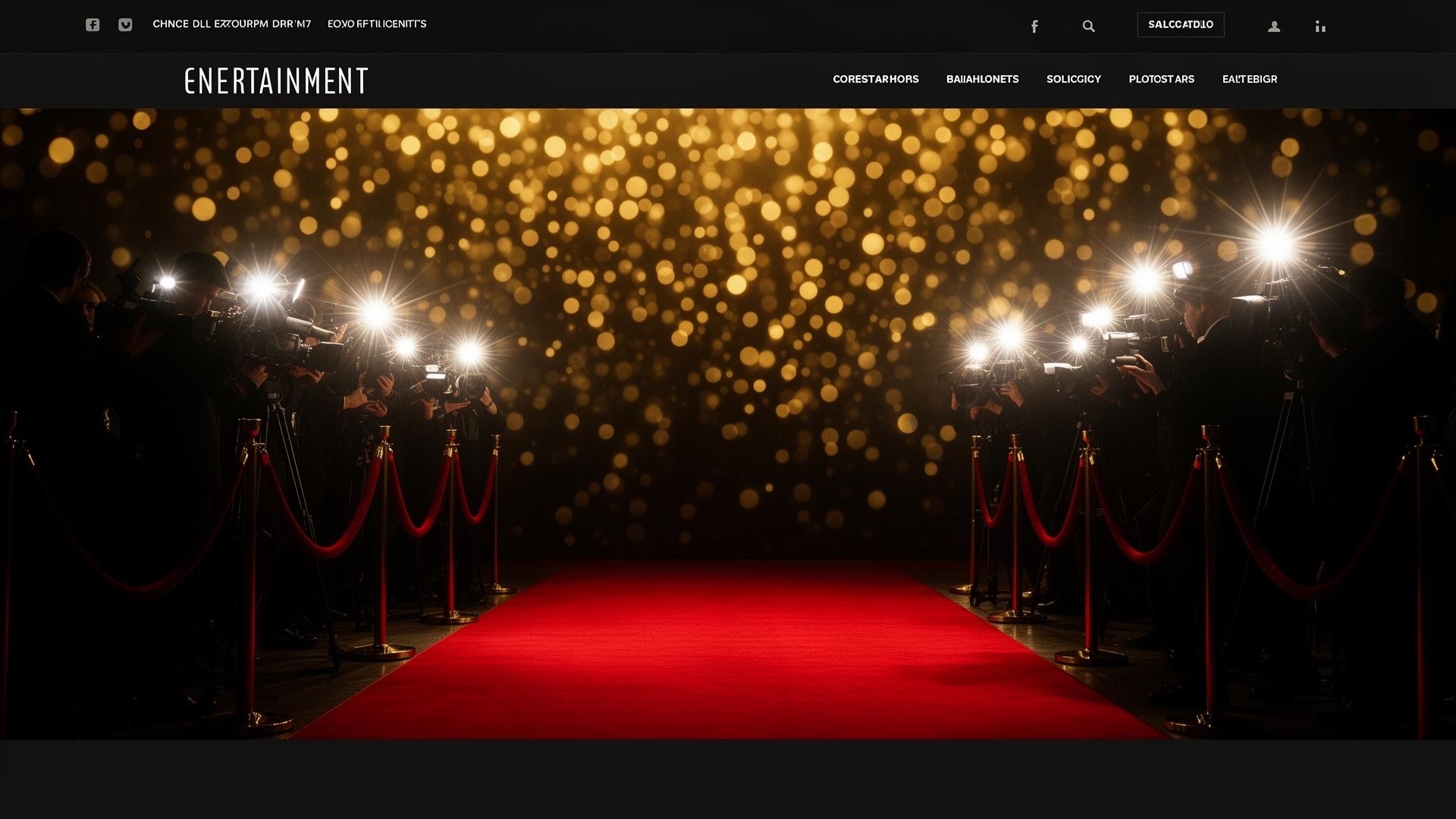
Exclusive Celebrity Photos
& Entertainment
Explore thousands of HD celebrity photos, red carpet galleries, portraits, and behind-the-scenes moments from Hollywood's biggest stars.
Explore Categories
Browse our curated celebrity photo collections
Celebrity Net Worth
Discover how much your favorite celebrities are worth
Actors
Singers
Rappers
Rock Stars
Comedians
Latest Articles
Celebrity news, photo galleries, and entertainment updates
Your Source for Celebrity Photos & Entertainment
PhotosCelebrities is your premier destination for high-quality celebrity photos, exclusive red carpet galleries, and entertainment content. We curate thousands of celebrity images from photo shoots, events, premieres, and behind-the-scenes moments.
Browse our extensive collection of female celebrity photos, celebrity portraits, paparazzi shots, and iconic celebrity pictures. From Hollywood actresses to Bollywood stars, our galleries cover the entire entertainment industry with daily updates.
Discover famous celebrity quotes, explore celebrity homes, and relive iconic moments with our then-and-now photo comparisons. PhotosCelebrities brings you closer to the stars you love.











